There is a new way to generate electricity from seawater

Scientists have used sunlight to turn seawater into hydrogen peroxide (H2O2), which can then be used in fuel cells to generate electricity
Scientists have used sunlight to turn seawater into hydrogen peroxide (H2O2), which can then be used in fuel cells to generate electricity
The solar energy sector is growing fast, despite the large fluctuations in the energy produced depending on the length of the daytime. This disadvantage could be overcome, however, when solar power is stored in the form of chemical energy and used as a fuel to produce electricity. A team of researchers, led by Shunichi Fukuzumi at Osaka University, has developed a method of photocatalytic production of hydrogen peroxide (H2O2) from seawater, which can subsequently be used to generate electricity in fuel cells. Their work was recently published in the journal Nature Communications.
Up to now, H2 -produced by photocatalytic pure water splitting- is typically used in fuel cells to generate electricity. However, H2 production has low solar energy conversion efficiency and due to its gaseous nature, it is difficult to store and transport. H2O2, on the other hand, can also be used as a fuel, even though previous methods of producing it required too much energy. The Osaka team managed to produce H2O2 as an aqueous solution from water and O2 in the air, which is much easier and safer to store and transport in higher densities, compared to highly compressed hydrogen gas. This efficient photocatalytic method involves a new photoelectrochemical cell developed to produce H2O2 when sunlight illuminates the photocatalyst, which then absorbs photons and initiates chemical reactions with the energy, resulting in H2O2. A test conducted for 24 hours shows that the H2O2 concentration in seawater reached about 48mM (millimolar), compared to 2mM in pure water. The researchers discovered that chlorine in seawater helped enhance the photocatalytic activity and led to the unusually high concentration of H2O2, and they expect that the method’s efficiency can be further improved by using better materials in the photoelectrochemical cell. "In the future, we plan to work on developing a method for the low-cost, large-scale production of H2O2 from seawater," Fukuzumi said. "This may replace the current high-cost production of H2O2 from H2 (from mainly natural gas) and O2".
Source: Phys.org
Source: Phys.org
Want to read more like this story?
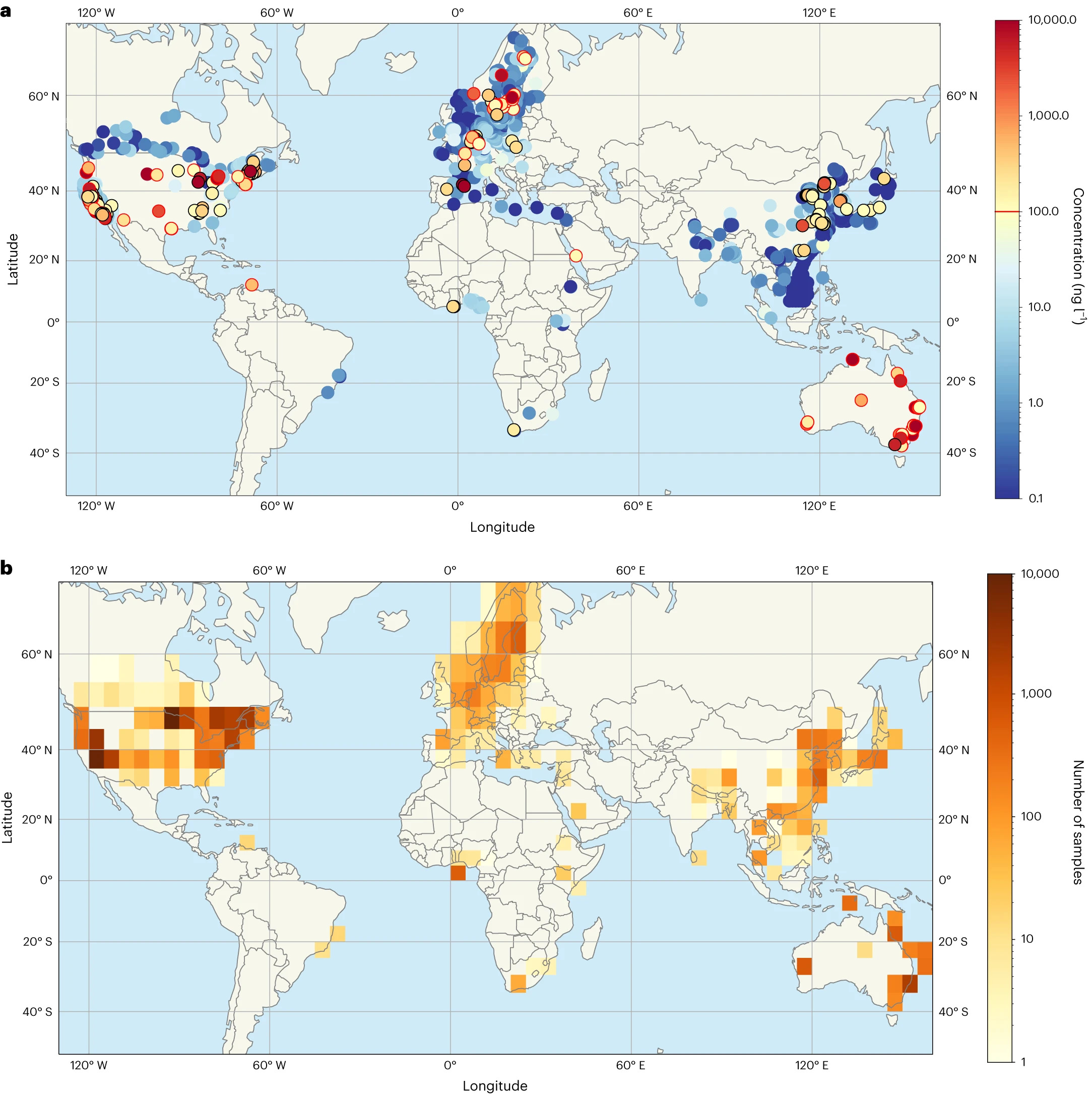
New study finds above-limit quantities of PFAS “forever chemicals” in water globally
Apr, 08, 2024A new study, led by the University of New South W...

Ireland plans to build Europe’s first ammonia-fueled power plant
Dec, 21, 2023Two energy companies, Centrica and Mitsubishi Pow...

Japan inaugurates world’s largest nuclear fusion reactor
Dec, 01, 2023The world’s largest nuclear fusion reactor was in...

Processed spent coffee grounds can enhance concrete strength
Nov, 20, 2023Researchers at the Royal Melbourne Institute of T...
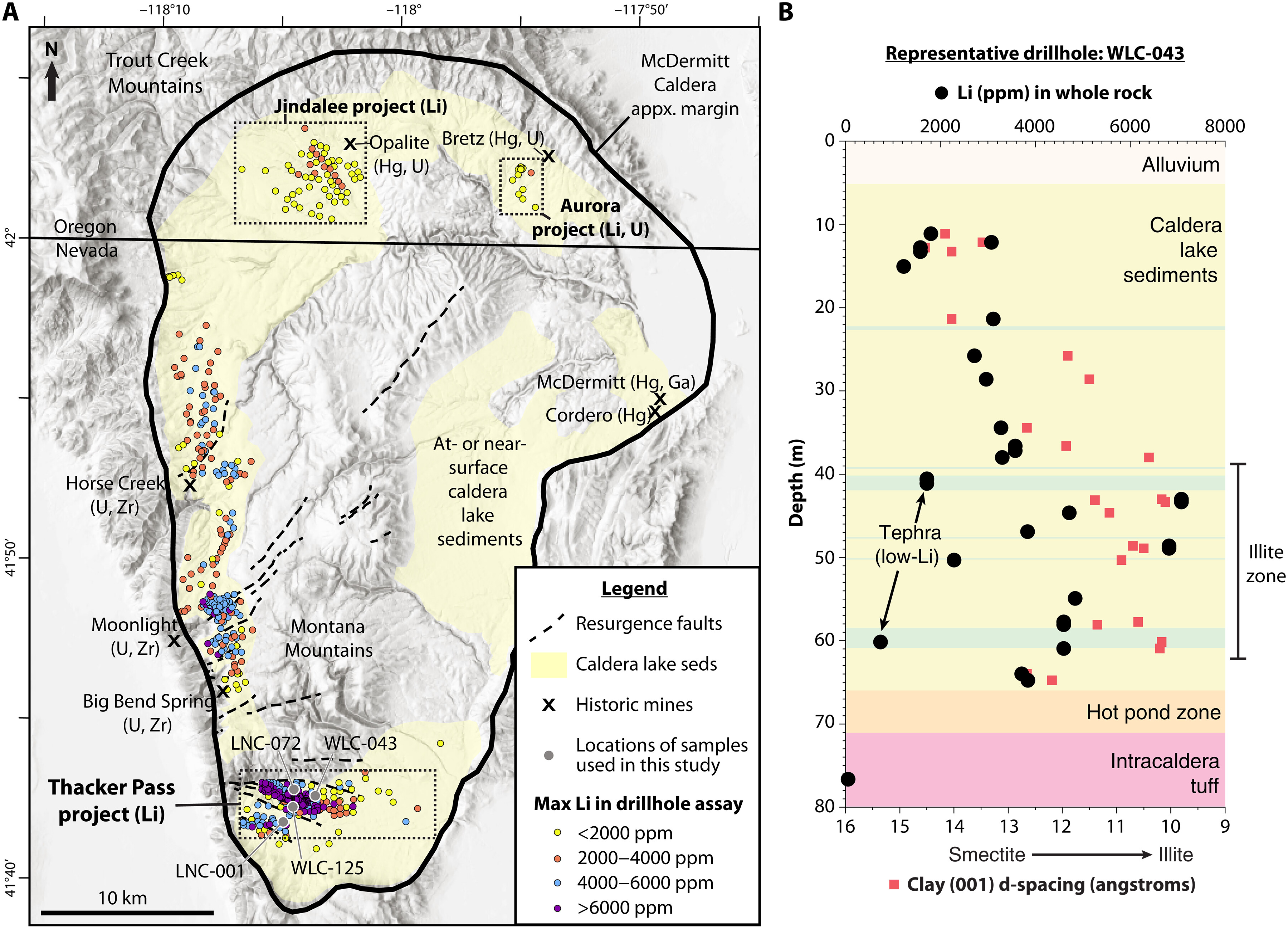
Potentially world’s largest known lithium deposit discovered in the US
Aug, 30, 2023A new study published on August 30, suggests that...

UN warns on the effects of sand dredging to the ocean floor and marine life
Sep, 05, 2023The UN Environment Programme (UNEP) warned on Tue...






